Last year, a student spilled water on a table and quickly wiped it with a sponge. The sponge soaked the water inside it.
Later in chemistry class, the same student saw charcoal cleaning dirty air by trapping gases on its surface.
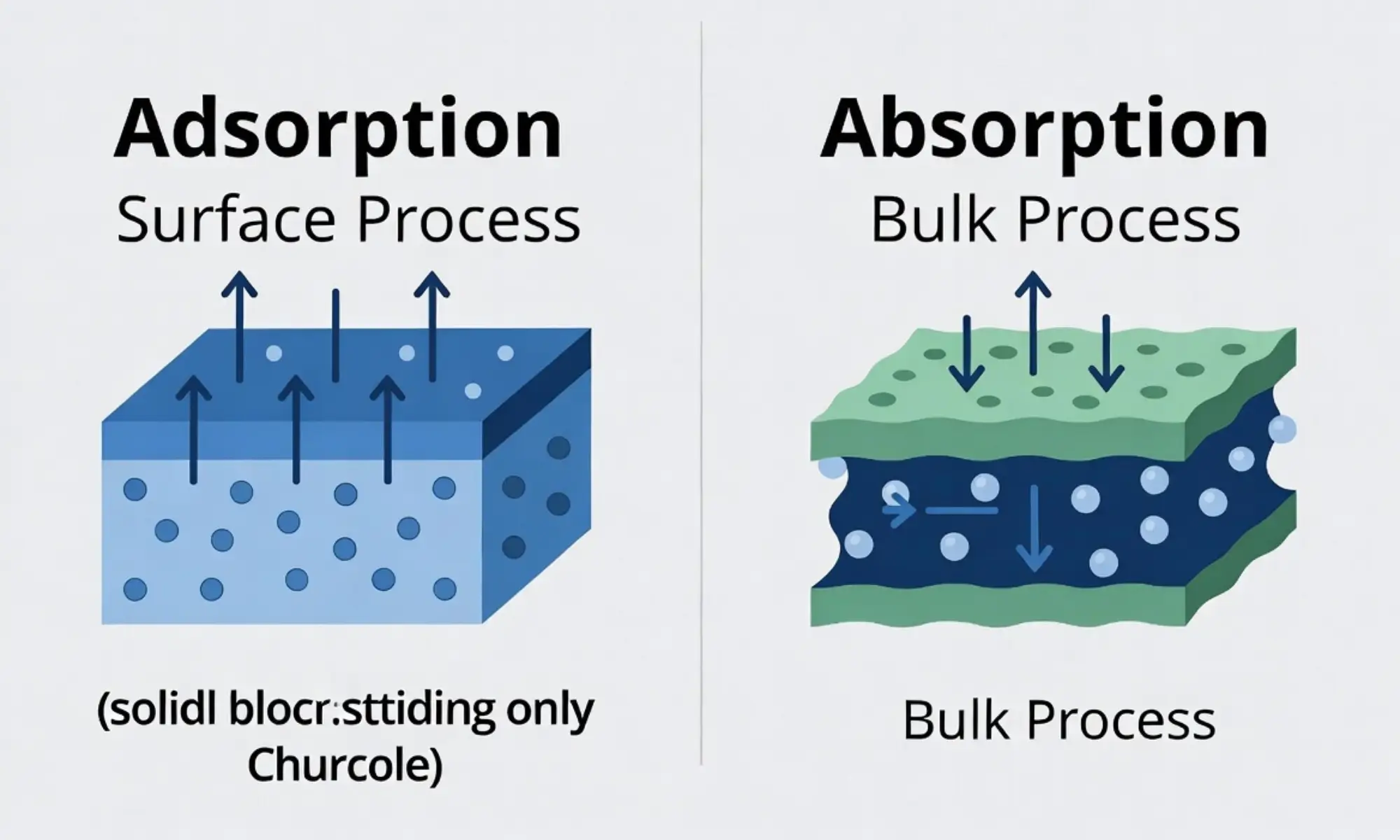
The teacher explained this was not soaking but sticking. That day, the student learned the difference between adsorption and absorption.
Absorption happens when a substance goes inside another substance. Adsorption happens when particles stick only to the surface.
Many learners confuse these two words because they sound alike. But the difference between adsorption and absorption is very important in science and daily life.
Understanding the difference between adsorption and absorption helps students, engineers, and doctors.
The difference between adsorption and absorption also plays a big role in industries and environmental protection.
Before we move ahead, let us first learn how to pronounce these terms correctly.
Pronunciation (US & UK)
- Adsorption
- US: /ədˈzɔːrp.ʃən/
- UK: /ədˈzɔːp.ʃən/
- US: /ədˈzɔːrp.ʃən/
- Absorption
- US: /əbˈzɔːrp.ʃən/
- UK: /əbˈzɔːp.ʃən/
- US: /əbˈzɔːrp.ʃən/
Key Difference Between the Both
The key difference is location.
- Adsorption happens on the surface.
- Absorption happens throughout the whole material (inside).
Why Is Their Difference Necessary to Know for Learners and Experts?
This difference is important in science and society. Chemists use adsorption to clean water and air. Doctors study absorption to understand how medicine enters the body. Engineers use both processes in manufacturing and environmental systems.
If learners confuse them, they may misunderstand chemical reactions. Experts must know the difference to design filters, medicines, and materials correctly. So, understanding this concept improves knowledge and supports real-world applications.
Now, let us explore the detailed differences.
Difference Between Adsorption and Absorption
Below are 10 simple points of difference.
1. Definition
- Adsorption: Sticking on the surface.
- Example 1: Gas sticking to charcoal surface.
- Example 2: Dust sticking to a wall.
- Example 1: Gas sticking to charcoal surface.
- Absorption: Soaking inside completely.
- Example 1: Sponge soaking water.
- Example 2: Cloth absorbing milk.
- Example 1: Sponge soaking water.
2. Process Type
- Adsorption: Surface process.
- Example 1: Paint sticking on wood surface.
- Example 2: Ink sticking on paper surface.
- Example 1: Paint sticking on wood surface.
- Absorption: Bulk process.
- Example 1: Water entering soil deeply.
- Example 2: Sugar dissolving in tea.
- Example 1: Water entering soil deeply.
3. Concentration
- Adsorption: High on surface only.
- Example 1: Gas layer on metal surface.
- Example 2: Moisture on outer layer.
- Example 1: Gas layer on metal surface.
- Absorption: Same throughout material.
- Example 1: Water evenly inside sponge.
- Example 2: Oil inside foam.
- Example 1: Water evenly inside sponge.
4. Rate
- Adsorption: Fast at first, then slows.
- Example 1: Charcoal quickly traps gas.
- Example 2: Filter works rapidly in start.
- Example 1: Charcoal quickly traps gas.
- Absorption: Steady process.
- Example 1: Sponge slowly fills up.
- Example 2: Medicine slowly enters blood.
- Example 1: Sponge slowly fills up.
5. Temperature Effect
- Adsorption: Decreases with heat.
- Example 1: Gas escapes from surface when heated.
- Example 2: Weak surface bonding at high temperature.
- Example 1: Gas escapes from surface when heated.
- Absorption: Increases with heat (often).
- Example 1: Sugar dissolves faster in hot tea.
- Example 2: Salt mixes faster in warm water.
- Example 1: Sugar dissolves faster in hot tea.
6. Examples in Nature
- Adsorption: Activated charcoal cleaning air.
- Example 1: Air purifiers.
- Example 2: Gas masks.
- Example 1: Air purifiers.
- Absorption: Plant roots absorbing water.
- Example 1: Roots taking nutrients.
- Example 2: Skin absorbing lotion.
- Example 1: Roots taking nutrients.
7. Energy Change
- Adsorption: Releases heat (usually).
- Example 1: Gas on metal surface.
- Example 2: Chemical bonding heat release.
- Example 1: Gas on metal surface.
- Absorption: May absorb or release heat.
- Example 1: Endothermic dissolving.
- Example 2: Heat exchange in solution.
- Example 1: Endothermic dissolving.
8. Reversibility
- Adsorption: Often reversible.
- Example 1: Gas removed by heating charcoal.
- Example 2: Surface cleaning possible.
- Example 1: Gas removed by heating charcoal.
- Absorption: Harder to reverse.
- Example 1: Dye absorbed in cloth.
- Example 2: Water fully inside sponge.
- Example 1: Dye absorbed in cloth.
9. Occurrence
- Adsorption: Solid surface + gas/liquid.
- Example 1: Metal + gas.
- Example 2: Carbon + vapor.
- Example 1: Metal + gas.
- Absorption: Liquid + liquid or solid + liquid.
- Example 1: Water + salt.
- Example 2: Oil + foam.
- Example 1: Water + salt.
10. Scientific Field
- Adsorption: Surface chemistry.
- Example 1: Catalysts in reactions.
- Example 2: Pollution control.
- Example 1: Catalysts in reactions.
- Absorption: Physical chemistry & biology.
- Example 1: Digestion process.
- Example 2: Medicine absorption in body.
- Example 1: Digestion process.
Nature and Behaviour
Adsorption behaves like a surface magnet. It attracts particles only outside.
Absorption behaves like a sponge. It takes particles deep inside.
Why Are People Confused?
Both words sound similar. Both involve taking in substances. The spelling difference is small, so learners mix them up.
Table: Difference and Similarity
| Aspect | Adsorption | Absorption | Similarity |
| Process | Surface | Bulk | Both involve particles |
| Location | Outer layer | Inside material | Physical/chemical process |
| Heat Effect | Decreases with heat | Often increases | Used in science |
| Example | Charcoal filter | Sponge | Used in daily life |
| Field | Surface chemistry | Biology/chemistry | Important in industry |
Which Is Better in What Situation?
Adsorption is better for cleaning and filtering.
Air purifiers use adsorption to trap harmful gases. Water filters use activated carbon to remove impurities. It is useful when we want to remove unwanted substances from the surface without mixing them inside.
Absorption is better for soaking and mixing.
In medicine, absorption allows drugs to enter the bloodstream. In agriculture, soil absorbs water to nourish plants. It is useful when full mixing or deep penetration is required.
Metaphors and Similes
- “He absorbed knowledge like a sponge.”
- “She adsorbed attention like a magnet.”
- “Ideas spread and were absorbed by the crowd.”
Connotative Meaning
Adsorption – Neutral technical term.
- Example: “Adsorption cleans polluted air.”
Absorption – Positive in learning.
- Example: “Her absorption in studies is inspiring.”
Idioms and Proverbs
- “Absorb the lesson.” (Learn deeply.)
- “Soak it in.” (Take fully inside.)
Example: During the lecture, students soaked it in carefully.
Works in Literature
- Absorption and Theatricality – Michael Fried (Art criticism, 1980)
- The Periodic Table – Primo Levi (Memoir, 1975)
Movies Related to Keywords
- Absorption (2009, USA – independent film)
- The Substance (2024, France/UK – science theme)
Five Frequently Asked Questions
1. Is adsorption same as absorption?
No. Adsorption is surface process; absorption is bulk process.
2. Which occurs in sponge?
Absorption.
3. Which is used in gas masks?
Adsorption.
4. Why is adsorption exothermic?
Because surface bonding releases heat.
5. Which is used in medicine?
Absorption allows drugs to enter blood.
How Both Are Useful for Surroundings
Adsorption cleans air and water.
Absorption helps plants grow and supports medicine.
Both protect environment and health.
Final Words for Both
Adsorption works on the surface. Absorption works inside. Both are powerful natural processes.
Conclusion
The difference between adsorption and absorption mainly lies in where the process happens. Adsorption takes place on the surface, while absorption occurs throughout the material.
Though they sound similar, their scientific meanings are different. Understanding this difference is important for students, scientists, and industries.
From cleaning air to absorbing medicine in the body, both processes are essential. Clear knowledge prevents confusion and improves scientific understanding.